BOSTON — Seaport Therapeutics said early-stage clinical data show its investigational drug GlyphAgo significantly improved bioavailability and reduced variability compared with standard agomelatine, supporting further development for generalized anxiety disorder.
Topline results from the Phase 1 proof-of-concept trial showed GlyphAgo achieved a 6.8-fold increase in bioavailability versus unmodified agomelatine, exceeding the company’s target of a twofold improvement. The drug also demonstrated a 10-fold reduction in pharmacokinetic variability, with no serious or severe adverse events reported.
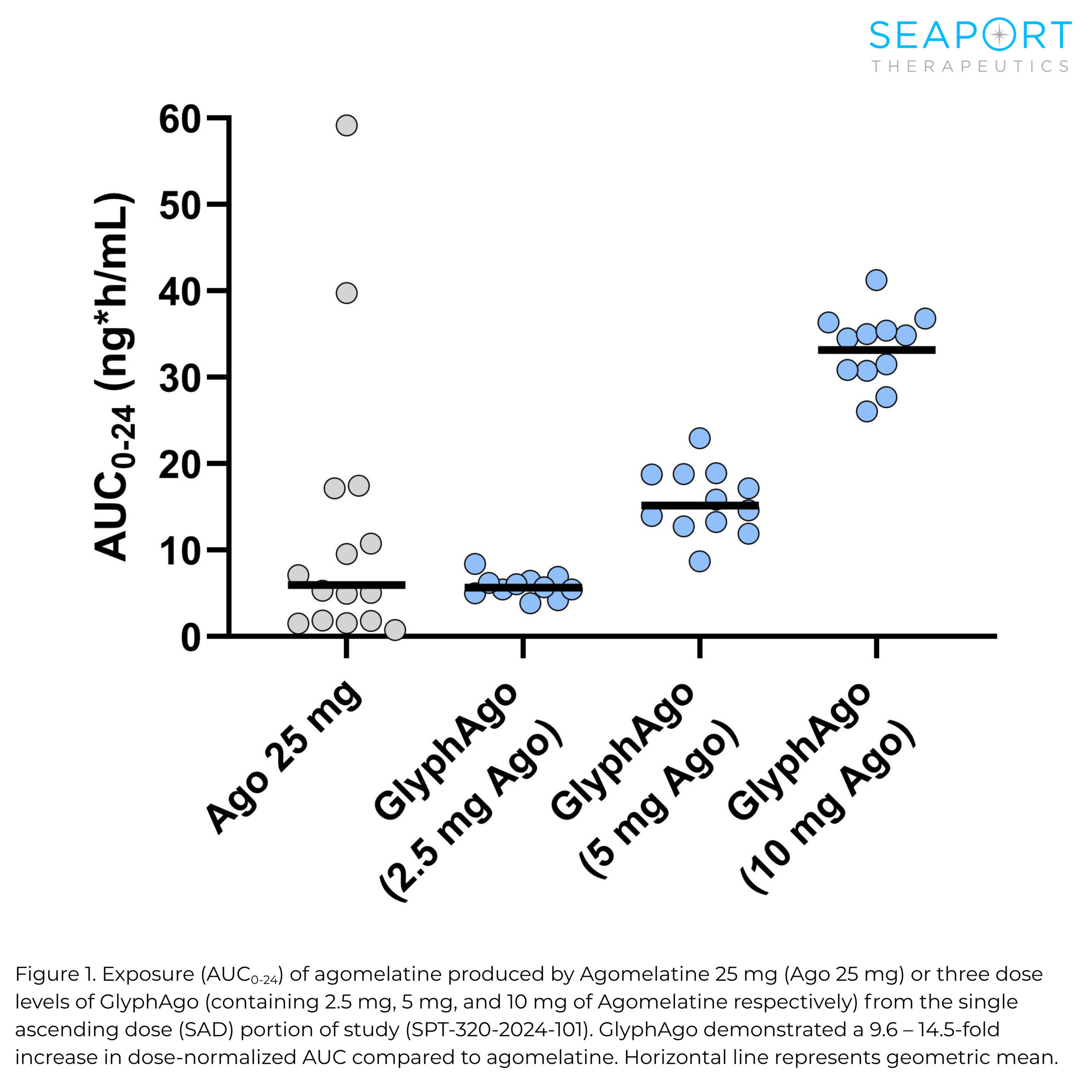 The study evaluated GlyphAgo, an oral prodrug of agomelatine, in healthy volunteers. Researchers said the drug achieved therapeutic exposure levels at lower doses, potentially reducing liver-related risks that have historically limited agomelatine’s use.
The study evaluated GlyphAgo, an oral prodrug of agomelatine, in healthy volunteers. Researchers said the drug achieved therapeutic exposure levels at lower doses, potentially reducing liver-related risks that have historically limited agomelatine’s use.
Daphne Zohar, co-founder and chief executive officer of Seaport Therapeutics, said the results validate the company’s platform and support advancing the program.
“These topline data, from a well-powered Phase 1 trial, strengthen our conviction in GlyphAgo’s potential and provide further clinical validation for the Glyph platform,” Zohar said. “Based on these data, we plan to advance GlyphAgo into two parallel trials, a Phase 2a proof-of-pharmacology trial to evaluate the potential sleep benefit of GlyphAgo in patients with GAD, and a Phase 2b trial in GAD, that is a randomized placebo-controlled trial designed to be registration-enabling. We believe that GlyphAgo has the potential to bring patients with generalized anxiety disorder what could be the first new therapy in 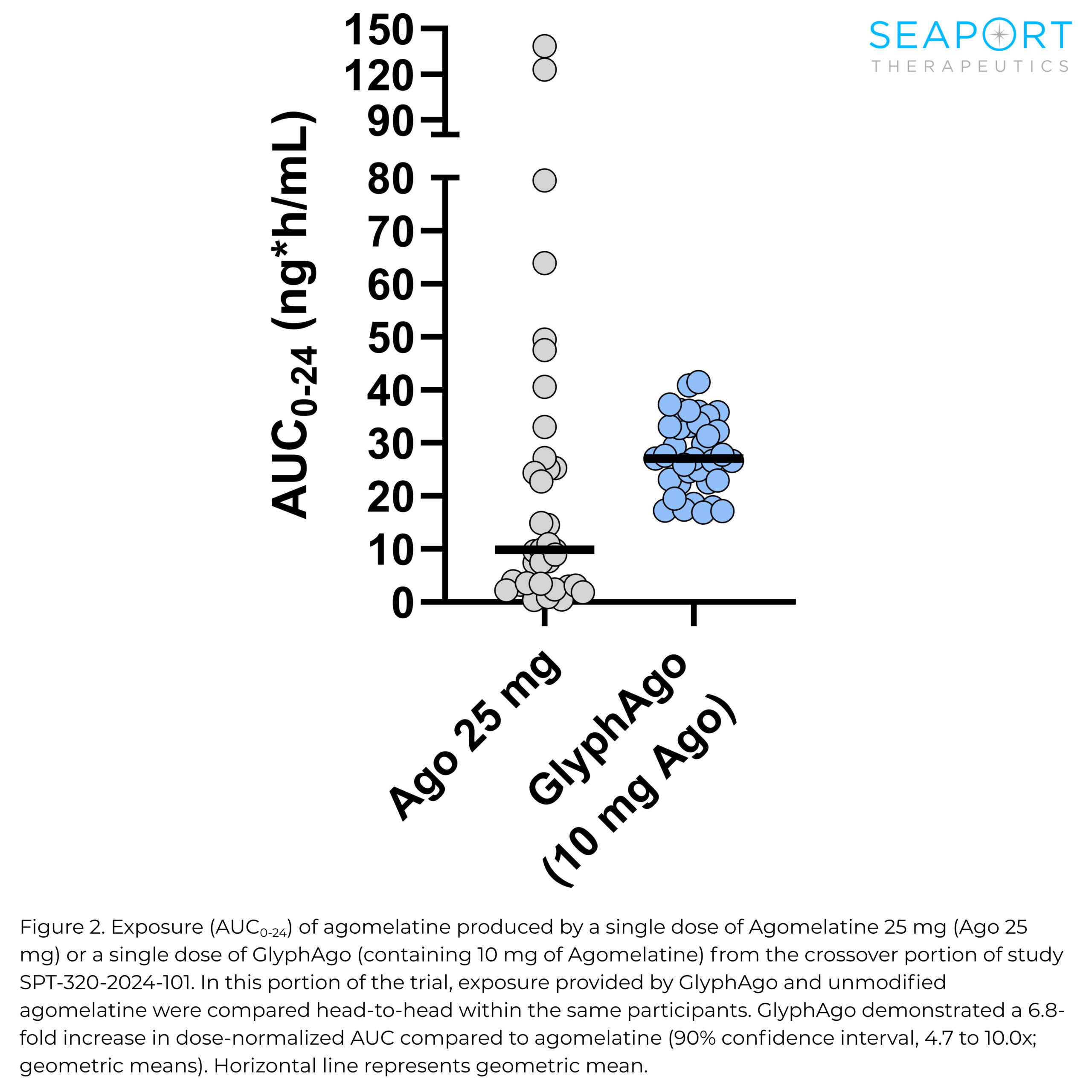 decades in the U.S. for this underserved and debilitating disorder.”
decades in the U.S. for this underserved and debilitating disorder.”
In addition to improved bioavailability, the drug showed consistent exposure levels regardless of factors such as food intake or use of estrogen-containing oral contraceptives, which are known to affect agomelatine metabolism. The trial included about 130 participants across single-ascending dose and crossover portions.
Dr. Steven Paul, co-founder and board chair of Seaport Therapeutics, said the findings address key limitations of existing therapies.
“In GAD, agomelatine has demonstrated robust and statistically significant separation from placebo in four third-party placebo-controlled studies and has been observed in meta-analysis to have better efficacy and tolerability than selective serotonin-reuptake inhibitors or benzodiazepines,” Paul said. “Despite this positive profile, over 90 percent of unmodified agomelatine is lost to first-pass metabolism and its use has been limited by dose-dependent liver enzyme elevations. The enhanced pharmaceutical properties of GlyphAgo and resulting markedly reduced inter-individual variability in systemic exposure to agomelatine support our clinical development of GlyphAgo in GAD.”
The company said GlyphAgo was designed to bypass first-pass liver metabolism by leveraging lymphatic absorption, increasing systemic exposure while reducing the need for liver function monitoring.
Seaport plans to present additional data from the ongoing trial, including results from the multiple-ascending dose portion, at upcoming scientific meetings.


