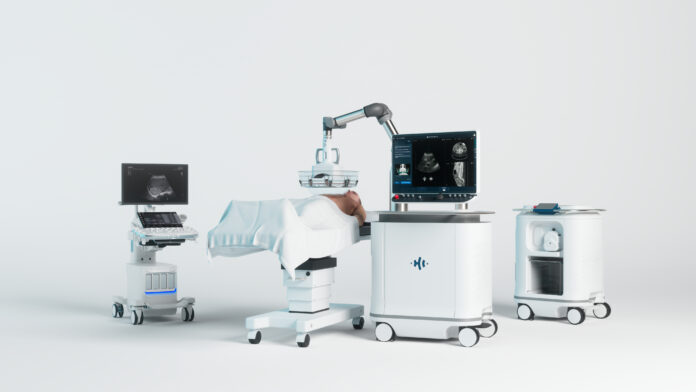
MINNEAPOLIS — HistoSonics has submitted a De Novo request to the U.S. Food and Drug Administration seeking authorization to expand use of its Edison Histotripsy System to include destruction of kidney tumors.
The company, which developed the Edison system and its histotripsy therapy platform, said the submission marks a step toward expanding the technology beyond liver tumors and into additional solid tumor applications.
Histotripsy uses focused ultrasound to mechanically liquefy and destroy targeted tissue without surgery, radiation or heat. HistoSonics said the approach could reduce the risk of complications and side effects associated with more invasive therapies.
“This submission is an important milestone in expanding histotripsy beyond the liver and into the kidney, an area where patients and physicians are seeking organ-preserving and nephron-sparing alternatives,” said Mike Blue, Chairman and CEO of HistoSonics. “We believe histotripsy has the potential to fundamentally change how solid tumors are treated, delivering precise, non-invasive therapy that preserves what matters most – including patient quality of life.”
The FDA submission is supported by clinical evidence from the ongoing #HOPE4KIDNEY trial, a prospective, multicenter, single-arm pivotal study evaluating the safety and effectiveness of the Edison system in destroying targeted kidney tumors. The trial enrolled 67 patients.
“Histotripsy represents a fundamentally novel approach to treating kidney tumors. In the #HOPE4KIDNEY Trial, we observed precise, noninvasive tumor destruction without incisions, radiation, or thermal injury. These findings highlight the potential of histotripsy to expand treatment options for patients with kidney tumors and represent an important step forward for both physicians and patients,” said Dr. William Huang, Urologic Oncologist at NYU Langone Health and Principal Investigator for the #HOPE4KIDNEY Trial.
HistoSonics said kidney tumors represent a growing clinical challenge, with about 680,000 people living with kidney tumors in the United States and an estimated 80,000 new cases expected to be diagnosed in 2026.
Current treatments for kidney tumors include partial nephrectomy and thermal ablation, which can carry risks such as bleeding, infection and unintended damage to surrounding tissue. HistoSonics said histotripsy could offer a non-invasive, non-thermal alternative designed to precisely target tumors while helping preserve kidney function.
The Edison system is currently indicated for non-invasive destruction of liver tumors, including unresectable liver tumors, using histotripsy. Its use in kidney applications remains investigational while under FDA review.