PRAGUE — H2 Global Group is positioning itself as a European technology platform linking capital with clinically validated healthcare innovation, citing strong investor returns and expansion plans tied to its portfolio company VR LIFE.
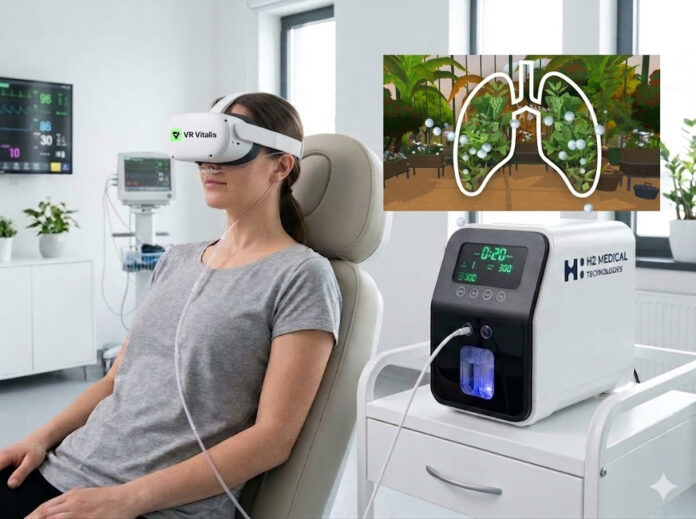
The company pointed to VR LIFE’s flagship MDR-certified medical device, VR Vitalis Pro, as a key example of its strategy. Following a recent $650,000 seed funding round backed by institutional investors, the implied value of H2 Global Group’s original investment in VR LIFE has more than doubled in less than three years to approximately $1.38 million.
VR LIFE is now preparing to seek U.S. Food and Drug Administration certification as it aims to enter the U.S. market, building on its clinical presence across more than 40 healthcare facilities in Europe.
H2 Global Group is also expanding its intellectual property portfolio through its subsidiary, H2 Medical Technologies. The company has developed a patent-pending therapeutic platform that combines molecular hydrogen with virtual reality for pulmonary rehabilitation. After receiving approval from the Czech State Institute for Drug Control in January 2026, the company has entered the registration phase for what it describes as the world’s first medical device using molecular hydrogen.
With a pre-money valuation of about $67 million, H2 Global Group said it is targeting a transaction value of between $500 million and $1.5 billion within the next three years.
“We are building a vertically integrated MedTech ecosystem that brings together breakthrough Japanese science, European clinical validation, and global capital to confront some of the most urgent healthcare challenges of our time,” said David Marsalek, CEO and founder of H2 Global Group. “We are advancing non-invasive solutions aimed at cognitive decline, Alzheimer’s disease, mild cognitive impairment, systemic inflammation, pulmonary rehabilitation, and the escalating quality-of-life crisis associated with aging populations.”
Marsalek added, “We are opening the door to U.S. and global partners who recognize this structural shift and want to capitalize on it.”
The company said its targeted therapeutic areas represent a combined addressable market exceeding $150 billion, including approximately $18 billion tied to Alzheimer’s disease and mild cognitive impairment and about $110 billion in anti-inflammatory therapeutics.